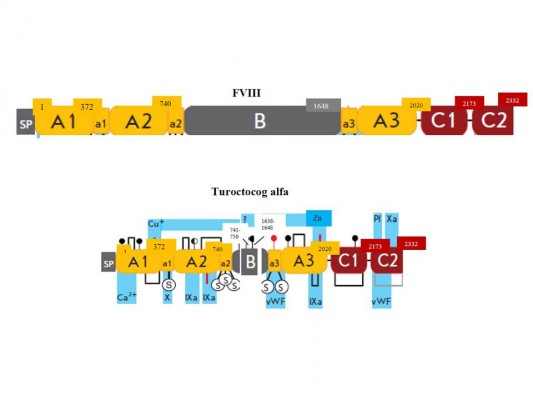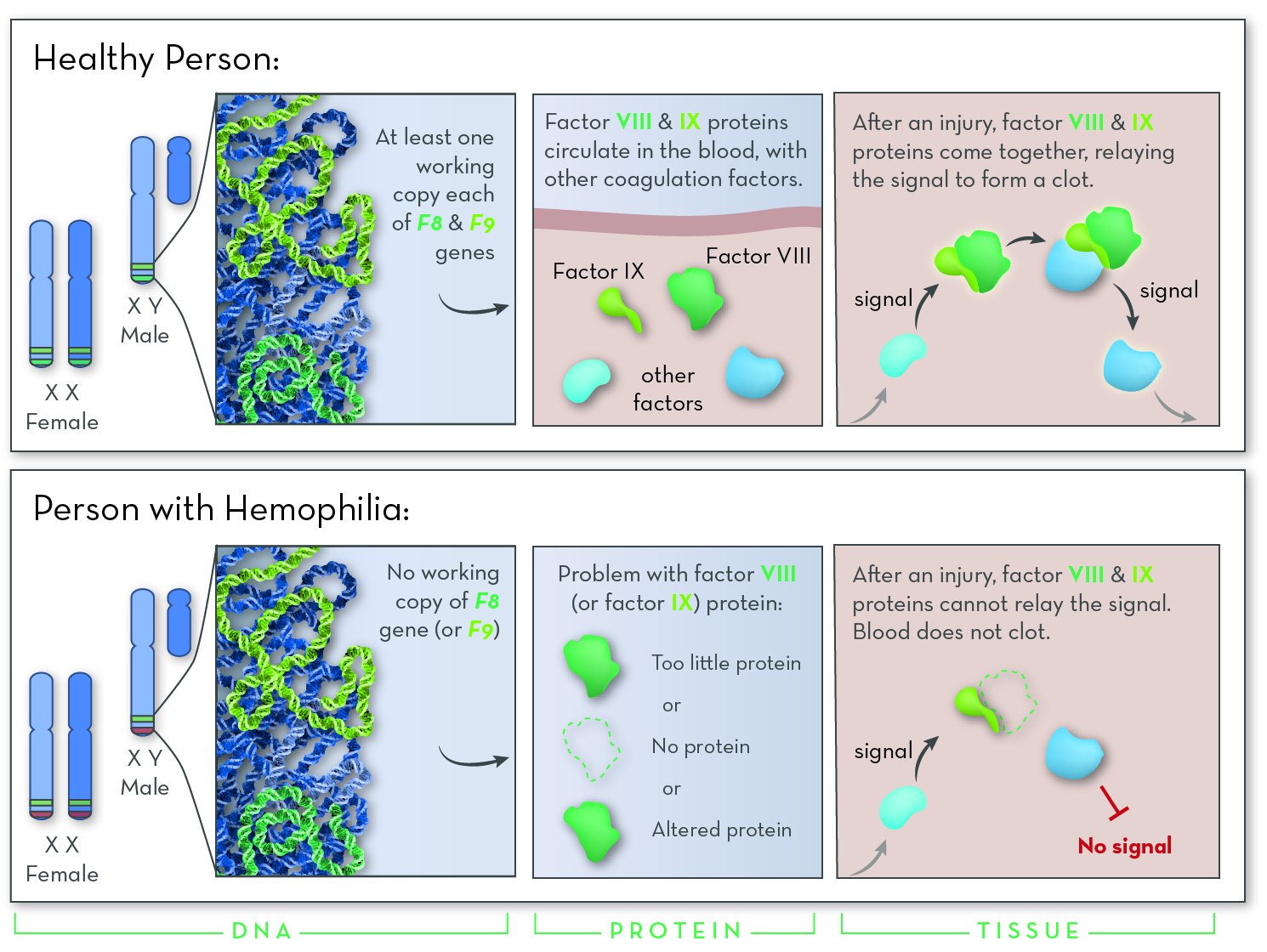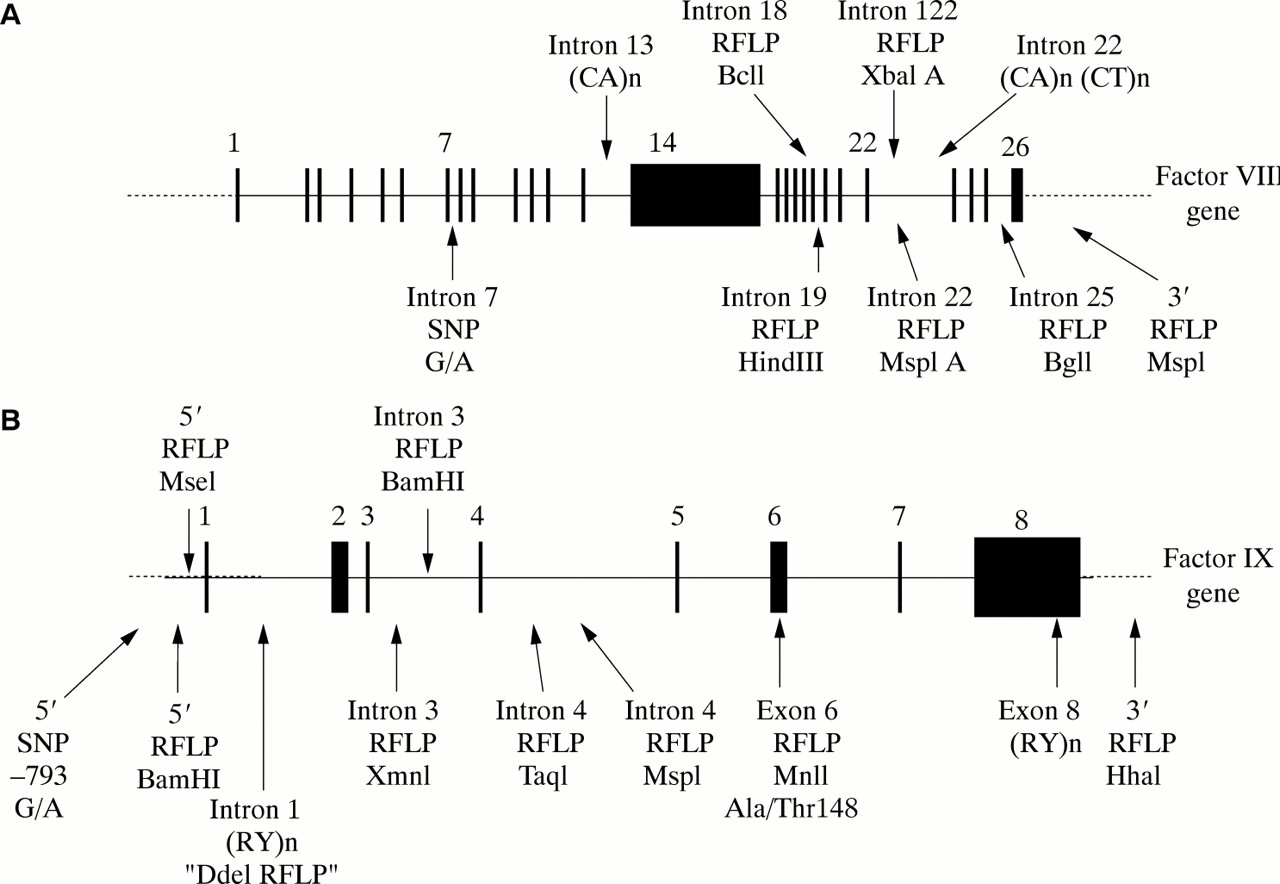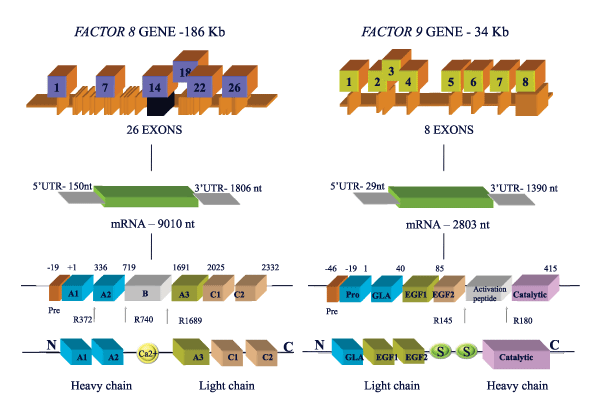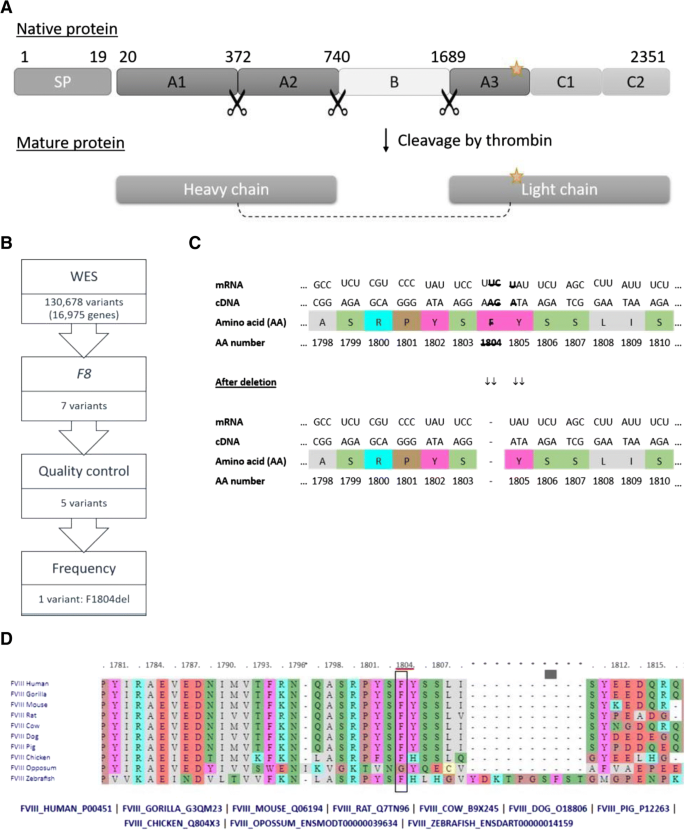
Identification of a novel mutation in the factor VIII gene causing severe haemophilia A | BMC Hematology | Full Text

NEJM on Twitter: "In this small study, AAV5–factor VIII gene transfer in severe hemophilia A was associated with normalization of factor VIII activity level at 1 year. https://t.co/R8DZso62ng https://t.co/smeflhHp65" / Twitter

Large deletion of FVIII (factor VIII gene): adapted from reference 35.... | Download Scientific Diagram

Large deletion of FVIII (factor VIII gene): adapted from reference 35.... | Download Scientific Diagram
![PDF] Factor VIII genetic mutations and protein alterations in hemophilia A: A review | Semantic Scholar PDF] Factor VIII genetic mutations and protein alterations in hemophilia A: A review | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7bcd3dd5b592881f7fe4b2e3160d24ea5148c6a0/5-Figure3-1.png)
PDF] Factor VIII genetic mutations and protein alterations in hemophilia A: A review | Semantic Scholar

Partial F8 gene duplication (factor VIII Padua) associated with high factor VIII levels and familial thrombophilia - ScienceDirect
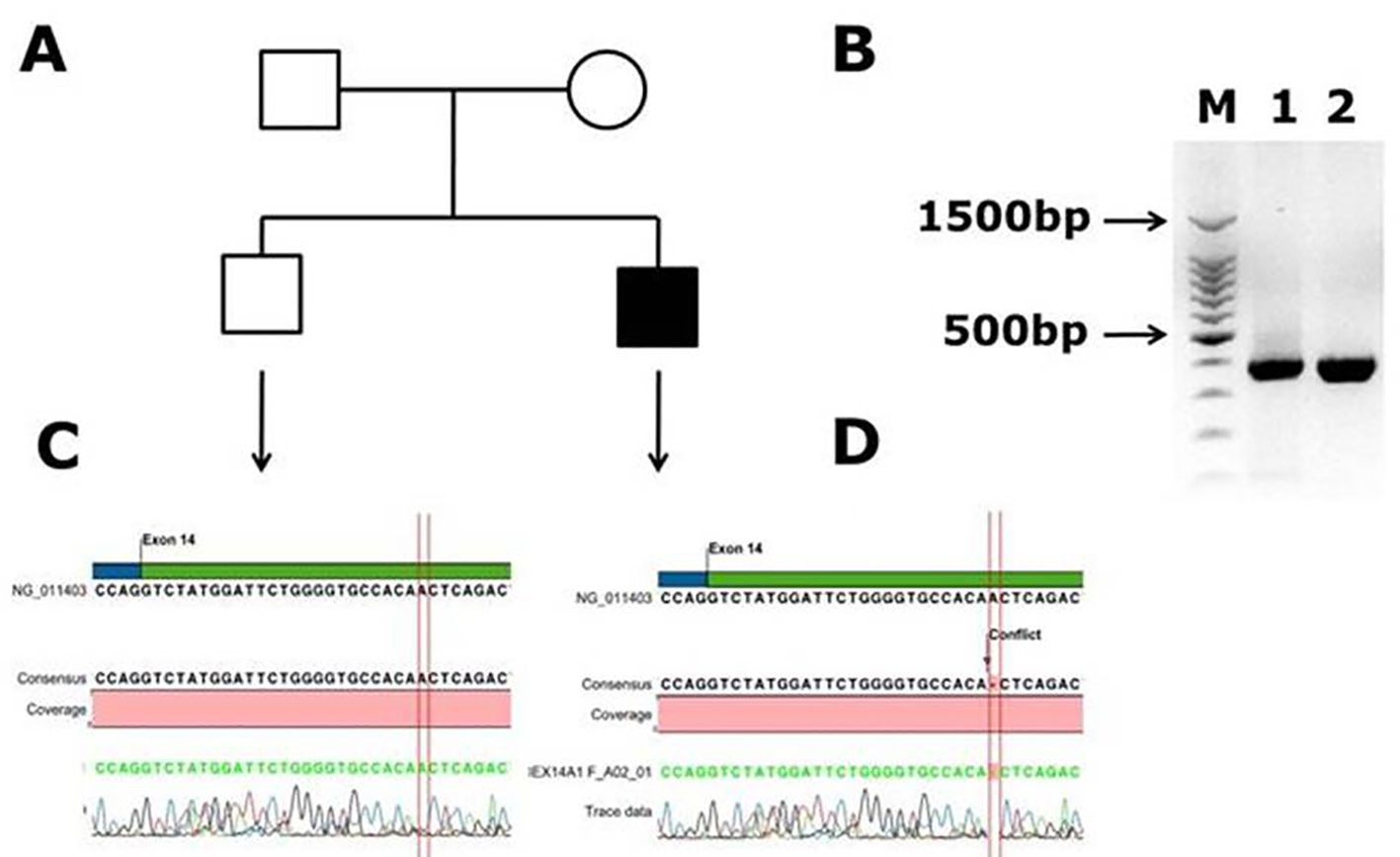
Molecular Analysis of Factor VIII and Factor IX Genes in Hemophilia Patients: Identification of Novel Mutations and Molecular Dynamics Studies | Al-Allaf | Journal of Clinical Medicine Research

Hemostatic Response is Maintained for up to 5 Years Following Treatment with Valoctocogene Roxaparvovec, an AAV5-hFVIII-SQ Gene Therapy for Severe Hemophilia A - ISTH Congress Abstracts
Efficacy and Safety of Valoctocogene Roxaparvovec Adeno-associated Virus Gene Transfer for Severe Hemophilia A: Results from the Phase 3 GENEr8-1 Trial - ISTH Congress Abstracts
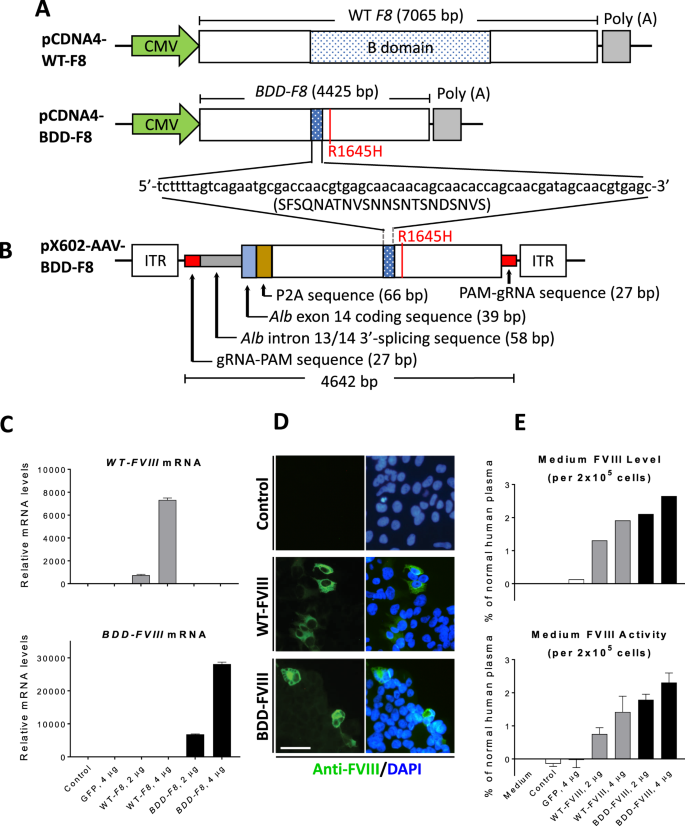
Hemophilia A ameliorated in mice by CRISPR-based in vivo genome editing of human Factor VIII | Scientific Reports